Keynote 048 results11/19/2023 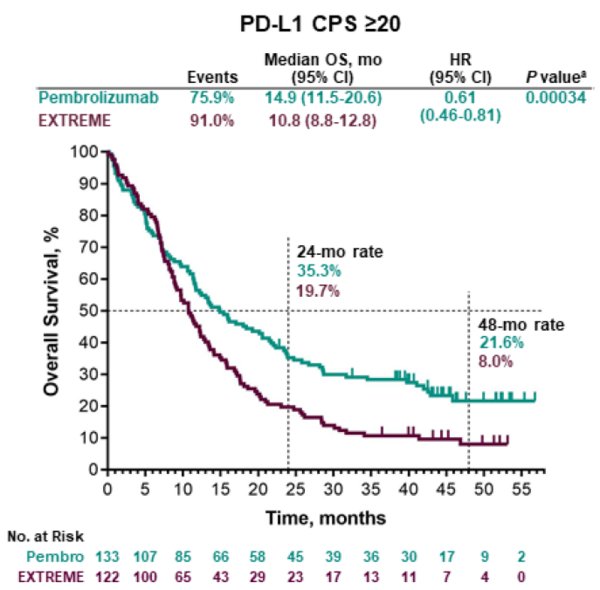
PD-L1 IHC 22C3 pharmDx Interpretation Manual – HNSCC. Presented at ESMO Virtual Congress 2020 19–21 September 2020. Presented at American Society of Clinical Oncology (ASCO) Annual Meeting 2020. KEYTRUDA Summary of Product Characteristics.= Number NR = Not Reported ORR = Objective Response Rate OS = Overall Survival P16 = Cyclin-Dependent Kinase Inhibitor 2A PD = Progressive Disease PD-1 = Programmed Cell Death-1 PD-L1 = Programmed Death Ligand-1 PFS = Progression-Free Survival PFS2 = Time From Randomisation To Objective Tumour Progression On Next-Line Treatment Or Death From Any Cause PR = Partial Response Q1W = Every Week Q3W = Every 3 Weeks Q6W = Every 6 Weeks QoL = Quality Of Life R = Randomised RECIST = Response Evaluation Criteria In Solid Tumours SCC = Squamous Cell Carcinoma TPS = Tumour Proportion Score TRAE = Treatment-Related Adverse Events. The phase 3 KEYNOTE-048 trial aimed to determine whether Keytruda alone or in combination with platinum-based chemotherapy improves overall survival (OS) compared with Erbitux plus platinum-based chemotherapy in participants with previously untreated recurrent or metastatic HNSCC that is not able to be cured by local therapies. Abbreviationsĥ-FU = 5-Fluorouracil AE = Adverse Event AEOSI = Adverse Events Of Special Interest AUC 5 = Desired Carboplatin Exposure of 5 mg/ml CI = Confidence Interval CPS = Combined Positive Score CR = Complete Response DoR = Duration Of Response ECOG PS = Eastern Cooperative Oncology Group Performance Status ESMO = European Society For Medical Oncology EXTREME = Cetuximab + 5-Fluorouracil + Platinum-Based Chemotherapy HNSCC = Head And Neck Squamous Cell Carcinoma HR = Hazard Ratio IHC = Immunohistochemistry ITT = Intention To Treat IV = Intravenous mo = Month M/uR = Metastatic Or Unresectable Recurrent No. KEYNOTE-048 trial was a multicentre, randomised, open-label, active-controlled Phase III study. Phase III KEYNOTE-048: Merck presented the first long-term results from the Phase III KEYNOTE-048 trial which evaluated KEYTRUDA monotherapy or in combination with chemotherapy for the first-line treatment of recurrent or metastatic head and neck squamous cell carcinoma. Conclusions: Pembrolizumab monotherapy and pembrolizumab-chemotherapy extended OS while maintaining HRQoL, further supporting first-line use for R/M HNSCC. *The KEYNOTE-048 study employed the PD-L1 IHC 22C3 pharmDx assay (Agilent Technologies, Carpinteria, CA, USA). PD-L1 testing supports a targeted treatment approach for all appropriate patients with M/uR HNSCC* 1 CPS ≥ 1 in the KEYNOTE-048 study 1 squamous cell carcinoma: results from the phase Ib KEYNOTE-012 expansion co. 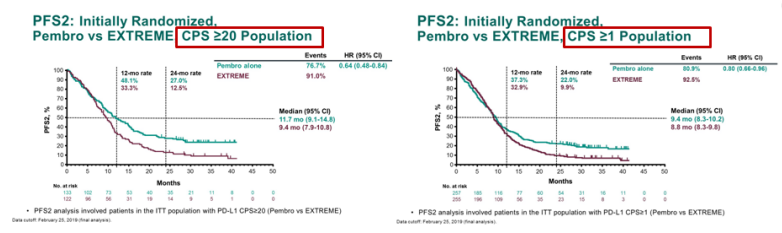
It concluded that 400 mg Q6W dosing regimen for KEYTRUDA monotherapy and combination is predicted to have a similar efficacy and safety profile as the approved 200 mg Q3W dosing regimen. Recently, a third phase III randomized trial. The study that led to the approval of the Q6W for monotherapy and combination patients assessed the 400 mg Q6W dosing schedule based on an exposure-response evaluation using modelling and simulation. Both doses are administered as IV infusions over 30 minutes
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
